Integrated quality support
When research environments cross regulatory boundaries
Many research programmes do not sit neatly within a single regulatory framework. Laboratories, clinical trials, and digital systems often intersect in ways that require coordinated oversight and thoughtful quality strategy.
Headway helps organisations navigate these complex environments by bringing together the right expertise across GLP, GCP and digital quality.
SENIOR-LEVEL SUPPORT
Research quality is rarely confined to one domain

Translational research programmes
Non-regulated discovery research that will inform GLP studies or contribute to investigator brochures.
Some of the most challenging quality environments arise when research moves between regulatory spaces or combines different scientific and operational disciplines.
These situations often require more than a single audit or compliance review. They require a joined-up understanding of how different parts of the research programme interact.

Laboratory research supporting clinical trials
Specialised laboratory analysis or biomarker testing linked directly to clinical trial outcomes.

Organisations moving into regulated research
Academic groups, laboratories or biotech companies conducting GCP or GLP work for the first time.

Digital systems embedded within research programmes
Clinical trial systems, laboratory platforms or analytical tools requiring CSV oversight within complex environments.

Clinical trials involving specialised laboratory platforms
Trials relying on non-standard laboratory technologies or distributed analysis environments.
strategic perspective
Why these environments are often difficult to support
Traditional consultancy approaches tend to treat regulatory environments separately. A GLP specialist reviews laboratory activities. A GCP specialist focuses on clinical trials. A CSV consultant evaluates digital systems.
While each perspective may be technically sound, the result can be fragmented oversight and disconnected recommendations.
Organisations are often left trying to reconcile multiple reports rather than receiving a clear strategy for the research programme as a whole.
a coordinated team approach
Headway works differently in complex environments
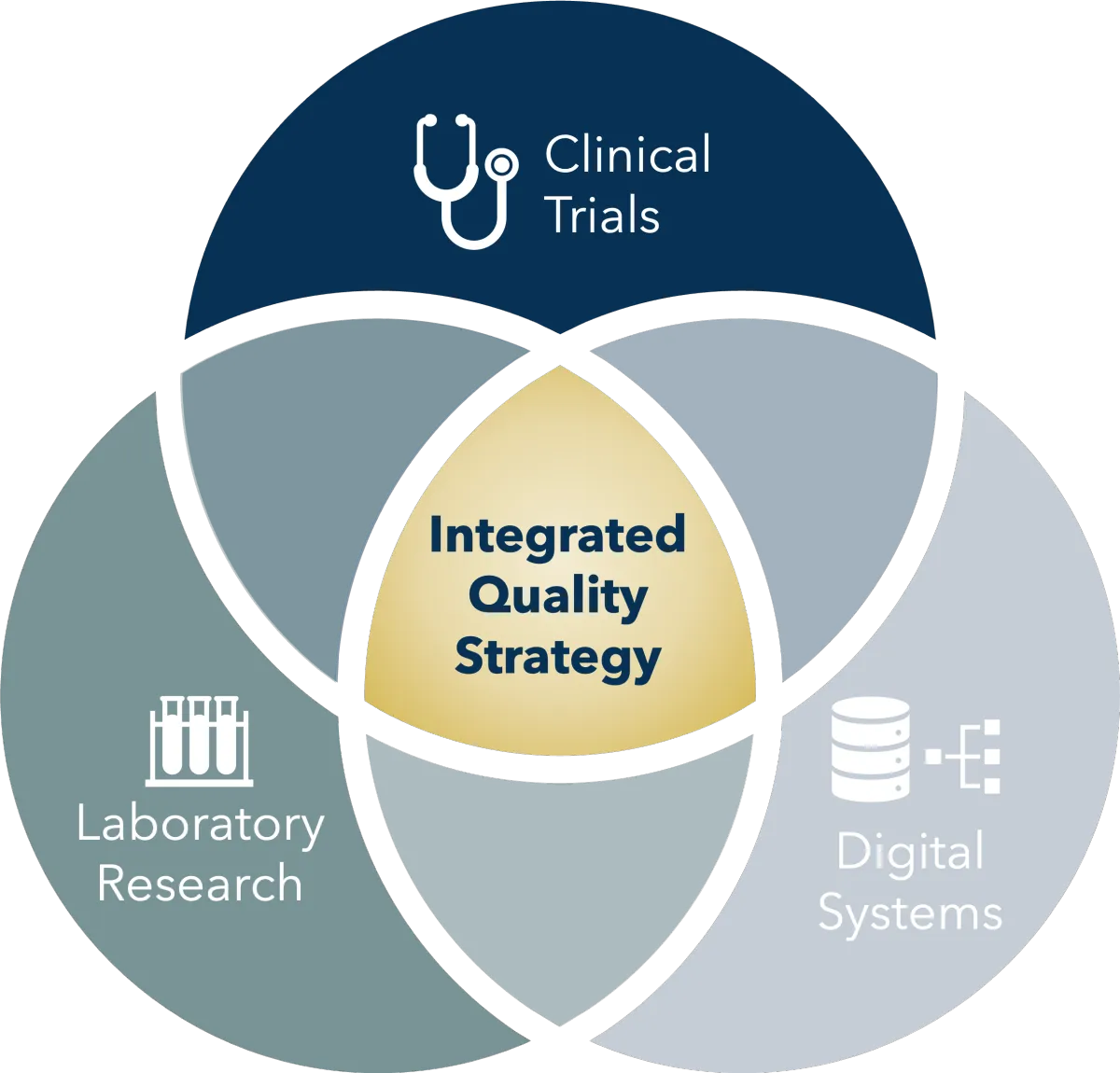
Rather than deploying isolated specialists, we bring together the expertise required to understand the full research context. Our consultants collaborate to examine how laboratory work, clinical research, digital systems and regulatory expectations interact.
This allows us to design integrated solutions that support the organisation’s scientific objectives while maintaining robust quality oversight.
A straightforward way to get started
A collaborative approach to complex challenges
1
Understand the research environment
We begin by understanding the full scientific and operational context of your research programme.
You gain an independent perspective on how the different parts of your research environment interact.
2
Define the outcome that matters
Together we clarify what confidence means for your organisation - whether that relates to regulatory readiness, research reliability, or decision-making support.
Quality oversight becomes aligned with the objectives of the research programme.
3
Bring together the right expertise
Headway assembles the appropriate combination of specialists across laboratory quality, clinical research quality and digital systems oversight.
You receive a coordinated approach rather than separate streams of advice.
4
Deliver integrated insight
Our team works collaboratively to provide clear findings and practical guidance that reflect the full research environment.
Leadership receives a coherent strategy rather than disconnected recommendations.
A straightforward way to get started
What organisations gain
Integrated quality oversight brings clarity to complex research environments.

Clear understanding of the research environment
A practical view of how laboratory work, clinical research and digital systems interact.
Coherent quality strategy
Joined-up recommendations that support the research programme as a whole.
Confidence in critical decisions
Leadership can move forward knowing the research and compliance approach is sound.
Stronger research governance
Quality systems designed to withstand regulatory scrutiny and support reliable science.
Discuss your research environment
If your organisation is navigating research across laboratories, clinical trials or digital systems, we would welcome a conversation about how best to support your programme.

Helping research organisations move beyond compliance – and make better decisions with clarity.
Start Here
Our Services
Education Law
Family Law
Employment Law
Cirminal Law
Insights & Company
Online Forum
City Councel
In House Booking
24/7 Support
© 2013-2026 Headway Quality Evolution - All Rights Reserved.
Contact
FAQ
Privacy Policy
